(Thoracic EndoVascular Aortic Repair)
Understanding and Anticipating Complications in Aortic Repair
Learning Objectives
- Describe the types of aortic pathology that may be appropriately repaired by TEVAR
- Explain the monitoring techniques and principles that are useful for perioperative management of patients undergoing TEVAR
- Identify complications that may occur in the procedural suite or immediately post-procedure, and formulate a management plan
Case Description
18236489
A 72 year old male is being transferred in for TEVAR. He is known to the vascular surgery service and has a history of previous endovascular aortic repair (EVAR) for an infrarenal abdominal aortic aneurysm (AAA), hypertension on amlodipine and metoprolol, non-obstructive coronary artery disease (CAD) maintained on daily baby aspirin, and chronic kidney disease (CKD). He presented to an outside hospital with hypotension and double vision, and chest CT showed extensive aortic disease with proximal descending aneurysm >9cm and possible extravasation at the level of the subclavian artery.
On arrival, his hemodynamics are stable (HR 70s SR on esmolol infusion and BP 110s/60s on nicardipine infusion), he is grossly neurologically intact, and painfree. Repeat chest CT confirms aneurysmal disease without aortic dissection. The proximal descending thoracic aorta measures 9.6cm (enlarged from 6.3cm since the last scan in 2011) with distal descending thoracic aorta measuring about 3 cm in diameter. There is no evidence of contrast extravasation to suggest aortic rupture. There is a small left sided pleural effusion. An aortobiiliac stent is present without evidence of endoleak or thrombosis.
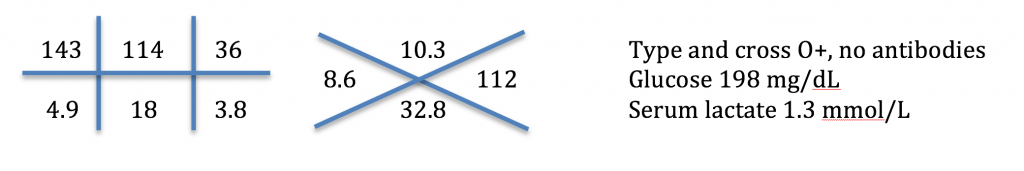
Admission lab studies are shown below:
Questions
How is an aortic dissection different from an aortic aneurysm?
- What is the significance of the pleural effusion found on CT?
- What patient or pathology characteristics may necessitate emergent TEVAR instead of an elective approach? What about selecting for open versus endovascular approaches to aortic repair?
Case Continues
The patient has arrived to the preop area. You find an anxious looking man, older than stated age, with family at bedside. Hemodynamics are unchanged from above, measured by NIBP on right arm. SpO2 98% on room air. He has multiple small gauge peripheral IVs in his upper extremities which are connected to the esmolol and nicardipine infusions. The surgeon says the procedure is not emergent but he would like to proceed with TEVAR today and there is a cath lab available.
Questions
- What monitors will you select for this patient? Are there any other monitoring choices or invasive procedures that you should discuss with your patient prior to anesthesia?
- Would your monitoring choices or anesthetic plan be different if the patient had been on dual antiplatelet agents (DAPT) or a novel oral anticoagulant (NOAC) leading up to admission?
- Does it matter which side you choose for your invasive blood pressure line?
Case Continues
After confirming that the patient is only on aspirin 81mg PO daily with no other anticoagulants or antiplatelet agents, and has normal baseline coagulation studies, you proceed to the cath lab. You place a right radial arterial line then a lumbar cerebrospinal fluid (CSF) drain. After a stable induction and intubation, additional right arm peripheral IV access is obtained and connected to a fluid warmer with a balanced salt solution. A triple lumen catheter is placed in the right internal jugular vein. The patient is positioned for surgery with the left arm on the surgical field, and the case proceeds.
Questions
- Why do the surgeons require the left arm to be available for some endovascular aortic repairs?
- What are your hemodynamic goals at this stage, and how does this change as the case progresses and grafts are deployed? What vasoactive substances would you have available to meet these goals?
- What can you do to mitigate risk of further renal injury?
- What is the role for transesophageal echocardiography in these cases?
Case Continues
Patient remains hemodynamically stable as the surgeon gets access and you titrate off the esmolol and nicardipine infusions. Patient receives 2 x 500mL crystalloid bolus with an improvement in urine output to >0.5mL/kg/hr. With fluid resuscitation and bleeding from around access sheaths, hematocrit value drifts toward 21%. You decided to transfuse 2 units PRBCs as the surgeons estimate at least 1-2 hours before the grafts are fully deployed. ACT is maintained >250 seconds with intermittent boluses of heparin. CSF drainage is clear, draining 5-10mL/hr with an ICP of 9, zeroed at the iliac crest. As grafts are deployed, the surgeon requests mean arterial pressure (MAP) to be driven up to 90-100mmHg. Angiography after final graft deployment shows no endoleak and surgeons remove access sheaths with good hemostasis.
Questions
- 1. Why did the surgeons request for the BP to be increased?
- 1. Why is it imperative for the patient to emerge from anesthesia as quickly and smoothly as possible?
- 1. What is an endoleak?
Case Continues to ICU
The patient is extubated after return of adequate spontaneous ventilation, reversal of neuromuscular blockade and demonstration of capacity to follow commands. He moved his upper extremities spontaneously and symmetrically. He is falling asleep when you try to examine his lower extremities prior to leaving the cath lab. On arrival to the ICU his HR is 80s sinus rhythm, MAP 130s/70s (MAP 80s). He is more awake and appears unable to move his legs. However, he states that he can feel normal sensation on all four extremities.
Questions
- What is going on?
- What needs to be done?
- How may this presentation differ from a neuraxial hematoma?

